There is currently no single solution that is better than another for all situations, but lithium-ion systems offer better cost of ownership in a number of situations, specifically hot climates, despite the higher initial cost.
Greg Albright, Jake Edie, Said Al-Hallaj | AllCell Technologies LLC
-
Introduction
.png) A wide variety of energy storage options are available today for the stationary power market; capacitors, compressed air, pumped hydro, flywheels and rechargeable batteries are all vying for a stake in the emerging role of energy storage. Each technology has its own merits based on a variety of application specific factors. This paper will focus on the comparison of two battery chemistries: lead acid and lithium-ion (Li-ion). The general conclusion of the comparison is that while the most cost effective solution is dependent upon a number of factors, there is a large market segment where lithium-ion has a lower cost of ownership when compared to lead acid. The figure below shows eleven variables that are included in the decision of what battery type to select for a given system.
A wide variety of energy storage options are available today for the stationary power market; capacitors, compressed air, pumped hydro, flywheels and rechargeable batteries are all vying for a stake in the emerging role of energy storage. Each technology has its own merits based on a variety of application specific factors. This paper will focus on the comparison of two battery chemistries: lead acid and lithium-ion (Li-ion). The general conclusion of the comparison is that while the most cost effective solution is dependent upon a number of factors, there is a large market segment where lithium-ion has a lower cost of ownership when compared to lead acid. The figure below shows eleven variables that are included in the decision of what battery type to select for a given system.

Figure 1: Battery Design Considerations
Within the scope of off-grid renewable systems, lead acid and nickel based batteries currently dominate the industry. Nickel batteries (NiCd, NiMH) are being phased out due to a combination of cost and environmental factors. Lead acid has been around for over 100 years and will be a market force for the foreseeable future due to its low cost and established manufacturing base. Lithium-ion is a well established technology for portable electronics but is still finding its role in larger scale applications; it is emerging as a contender in certain stationary applications where volume, weight, temperature sensitivity or low maintenance is more important than initial cost. The following chart illustrates how lead acid and lithium-ion fit into the rechargeable battery world.

Figure 2: Rechargeable Battery Types
-
Basics of Batteries
2.1 Basics of Lead Acid
Lead acid batteries have been around for more than a century. In the fully charged state, a 2V electric potential exists between the cathode and the anode. During discharge, electrons are passed externally through the load while internal chemical reactions at the interface of the electrolyte and the electrodes work to balance the charge equilibrium. Figure 3 illustrates the chemical states of a fully charged and discharged lead acid battery.

Figure 3: Lead Acid Charge States
Lead acid batteries can be divided into two distinct categories: flooded and sealed/valve regulated (SLA or VRLA). The two types are identical in their internal chemistry (shown in Figure 3). The most significant differences between the two types are the system level design considerations. Flooded lead acid batteries require three things that VRLA don’t:
- Upright orientation to prevent electrolyte leakage
- Ventilated environment to diffuse gases created during cycling
- Routine maintenance of electrolyte
Due to these differences, the lower cost of flooded lead acid must be balanced against the added complexity and secondary costs. VRLA batteries are divided into two categories: Gel and Absorbed Glass Mat (AGM). The different names reflect different methods of containing the electrolyte. In Gel batteries, a thickening agent is added to turn the electrolyte from liquid to gel. In AGM cells, a glass matrix is used to contain the liquid electrolyte.
“Deep cycle” and “shallow cycle” lead acid batteries can be found in both the VRLA and flooded classes. Shallow cycle VRLA batteries are commonly used for automotive start, light, ignition (“SLI”) batteries that must deliver high power pulses for short durations. The stationary power market uses deep cycle since the batteries will often discharge at a low rate over the course of multiple hours.
2.2 Basics of Lithium-ion
The concept of a lithium-ion battery was initially conceived in the 1970’s and began to see widespread adoption by the 1990’s. The basic mechanism is that a charged lithium ion is shuttled back and forth between the cathode and the anode during charge and discharge. Figure 4shows a diagram of a LiCoO2 variation of the lithium-ion family.
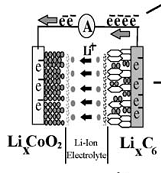
Figure 4: Lithium-ion reaction[i]
Chemistry differences in the cathode, anode, and electrolyte influence cell performance, as does packaging geometry. The cathode chemistry is the factor most commonly altered from cell manufacturer to cell manufacturer with terms like LFP, NCM, NCA, Cobalt, and Manganese reflecting the cathode chemistry class. Over 90% of lithium-ion anodes are comprised of graphite; silicon and titanium based materials are occasionally used to get better life and power performance in exchange for significantly higher cost.
The electrolyte exists in liquid form, but for “lithium polymer” cells, the electrolyte is absorbed in a polymer membrane. This allows for cell manufacturers to use a pouch enclosure on the cell rather than the metal casing used when liquid electrolyte is present in cylindrical and prismatic shaped cells. Each of these variations influences the performance of a lithium-ion cell.
In spite of the various chemical variations, lithium-ion batteries can generally be separated into two groups: lithium iron phosphate (LFP, LiFePO4) and metal oxides (NCM, NCA, Cobalt, Manganese). Table 1outlines the differences between the two chemistry classes on a cell level. The values in the table reflect average values as there is variation in each class.
Table 1: Lithium-ion subcategory comparison
|
LFP |
LiNCM |
|
|---|---|---|
|
Voltage |
3.3 V nominal (2-3.6 V/cell) |
3.7 V nominal (2.7-4.2 V/cell) |
|
Energy Density |
300 Wh/L |
735 Wh/L |
|
Specific Energy |
128 Wh/kg |
256 Wh/kg |
|
Power |
1000 W/kg |
512 W/kg |
|
Cycle Life |
2,000 @ 100% DoD 3,000 @ 80% DoD |
750 @ 100% DoD 1,900 @ 80% DoD |
|
Calendar Life |
6 years |
8 years |
|
Max recommended temperature |
40°C |
55°C |
|
Safety |
High |
Moderate |
|
Commercial Suppliers |
A123, Valence, BAK, BYD, K2, Lishen, many Chinese vendors |
Sanyo, Panasonic, Samsung, DowKokam, Sony, LG Chem, Moli |
All lithium-ion cells are “deep cycle” meaning that they have the ability to be fully charged and discharged. The life of the battery will significantly increase if the depth of each discharge is limited to 80% of the rated capacity.
-
Comparing lithium-ion to lead acid
Table 2 provides a brief comparison of lead acid to lithium-ion (LiNCM) on a pack level. It should be noted that both chemistries have a wide range of parameter values, so this table is only a simplified representation of a very complex comparison.
Table 2: Battery Technology Comparison
|
Flooded lead acid |
VRLA lead acid |
Lithium-ion (LiNCM) |
|
|
Energy Density (Wh/L) |
80 |
100 |
250 |
|
Specific Energy (Wh/kg) |
30 |
40 |
150 |
|
Regular Maintenance |
Yes |
No |
No |
|
Initial Cost ($/kWh) |
65 |
120 |
600[1] |
|
Cycle Life |
1,200 @ 50% |
1,000 @ 50% DoD |
1,900 @ 80% DoD |
|
Typical state of charge window |
50% |
50% |
80% |
|
Temperature sensitivity |
Degrades significantly above 25°C |
Degrades significantly above 25°C |
Degrades significantly above 45°C |
|
Efficiency |
100% @20-hr rate 80% @4-hr rate 60% @1-hr rate |
100% @20-hr rate 80% @4-hr rate 60% @1-hr rate |
100% @20-hr rate 99% @4-hr rate 92% @1-hr rate |
|
Voltage increments |
2 V |
2 V |
3.7 V |
An interesting point in this table is that the different chemistries have different typical state of charge windows. The implication of this is that a lead acid system must have a larger nameplate energy capacity than the lithium-ion system to have the same amount of available energy.
Given the significant differences in technical and economic characteristics of the battery types, it stands to reason that the “best” solution for which battery type to use is application specific. Following is a more in-depth look at some of the topics addressed in Table 2.
3.1 Cycle Life Comparison
Lithium-ion has significantly higher cycle life than lead acid in deep discharge applications. The disparity is further increased as ambient temperatures increase. The cycle life of each chemistry can be increased by limiting the depth of discharge (DoD), discharge rate, and temperature, but lead acid is generally much more sensitive to each of these factors.
Figure 5 shows cycle life data for a lithium-ion pack compared to an AGM style VRLA battery in a moderate climate (average temperature of 77°F). As cycle life is influenced by depth of discharge, the figure shows multiple DoD percentages for the lead acid. It can be seen that the AGM pack must be limited to a 30% depth of discharge to get comparable life to a lithium-ion that is at 75% depth of discharge. This means that the AGM battery must be 2.5 times larger in capacity than the lithium-ion to get comparable life.
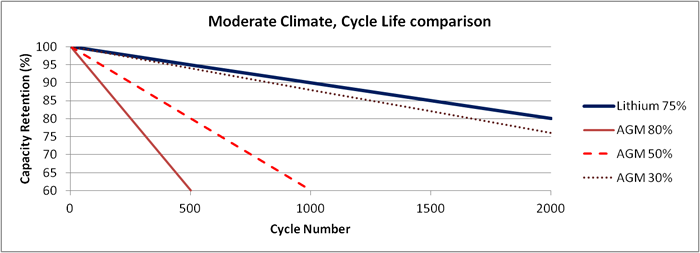
Figure 5: Cycle life, moderate climate
In hot climates where the average temperature is 92°F, the disparity between lithium-ion and lead acid is further exacerbated. The cycle life for lead acid (flooded and VRLA) drops to 50% of its moderate climate rating while lithium-ion will remain stable until temperatures routinely exceed 120°F. Figure 6 illustrates the disparity.
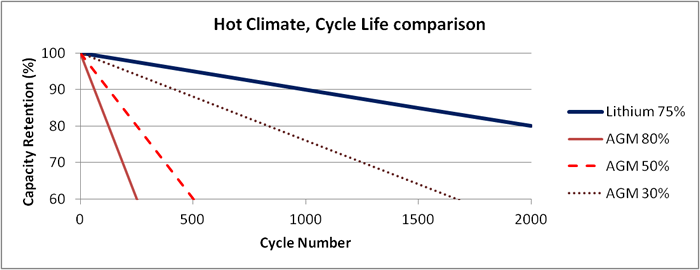
Figure 6: Cycle life, extreme climate
3.2 Rate Performance
When determining what capacity of battery to use for a system, a critical consideration for lead acid is how long the system will take to discharge. The shorter the discharge period, the less capacity is available from the lead acid battery.
A 100Ah VRLA battery will only deliver 80Ah if discharged over a four hour period. In contrast, a 100Ah lithium-ion system will achieve over 92Ah even during a 30 minute discharge. As shown in Figure 7, this condition makes lithium-ion very well suited for applications where full discharge occurs in less than eight hours.

Figure 7: Capacity vs. Discharge Rate
3.3 Cold Weather Performance
Both lead acid and lithium-ion lose capacity in cold weather environments, but as shown in Figure 8, lithium-ion loses significantly less capacity as the temperature drops into the -20°C range. The rate of discharge influences the lead acid performance, so two different rates have been shown for the VRLA battery.
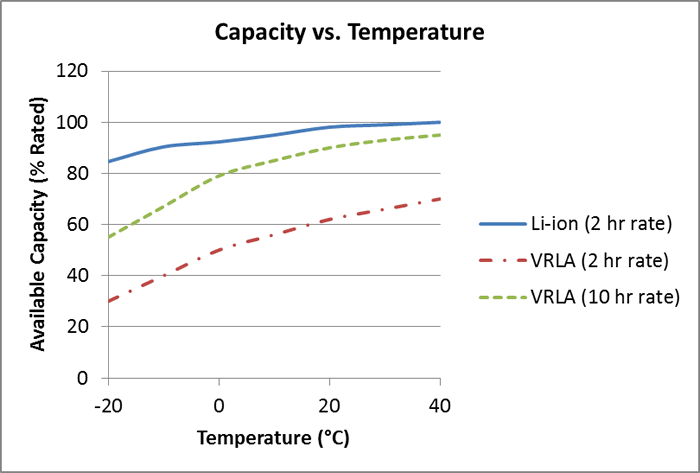
Figure 8: Capacity Available versus Temperature
3.4 Environmental Impact
Lead acid batteries compare poorly to lithium-ion with regards to environmental friendliness. Lead acid batteries require many times more raw material than lithium-ion to achieve the same energy storage, making a much larger impact on the environment during the mining process. The lead processing industry is also very energy intensive, leading to large amounts of pollution. Although lead is highly hazardous to human health, the manufacturing methods and battery packaging make the human risk negligible. On the plus side, over 97% of lead acid batteries in the United States are recycled, which makes a huge impact on the environmental equation.[ii]
Lithium is not without its own environmental problems.[iii] The major components of a lithium-ion cell require the mining of lithium carbonate, copper, aluminum, and iron ore. Lithium mining specifically is resource intensive, but lithium is only a minor portion of the battery cell by mass, so the aluminum and copper environmental impacts are much more significant. The lithium-ion recycling industry is only in its infancy right now, but the cell materials have shown high ability for recovery and recyclability, so it is expected that lithium-ion recycling rates will rival lead acid.
3.5 Safety
Lead acid and lithium-ion cells are both capable of going into “thermal runaway” in which the cell rapidly heats and can emit electrolyte, flames, and dangerous fumes. The likelihood and consequences of an event are higher for lithium-ion as it has a higher amount of energy in a smaller volume. Multiple cell and pack safety precautions shown in Figure 9 are taken to prevent trigger events, such as short circuits and overheating, but incidents still occur.

Figure 9: Lithium-ion safety mechanisms
3.6 Voltage Comparison
When evaluating if lithium-ion and lead acid can be interchangeable within a given electrical system, the most important factor is the voltage range of each chemistry. Figure 10 shows a comparison of three battery packs that are nominally called “24V” batteries. The LiNMC nominal voltage is technically 25.9V and the LFP is technically 25.6V.
The end result of the figure is that lithium-ion has good agreement with lead acid systems for a majority of the voltage range, but any electrical system would have to be able to accommodate the higher charging voltage of lithium-ion to get optimal performance. Most renewable energy battery charge controllers and discharge inverters are capable of being adjusted between lead acid and lithium-ion. Charge controller and inverter manufacturers and lithium-ion companies can assist in ensuring system compatibility.

Figure 10: Voltage comparison
-
Case Study
Given the variation of technical qualities between lead acid and lithium-ion, any given case study won’t necessarily apply to a broader range of applications. The system economics and result of the study are very sensitive to environmental conditions, available volume, charge/discharge rate, remoteness of installation and even local laws.
An emerging market where stationary energy storage is expected to play a significant role is the electrification of rural villages. The cost to run transmission lines is often prohibitive (>$1M/mile), so renewable systems with energy storage offer an attractive option. Coincidentally, many of the prospective installation sites are in warmer climates, which tips the balance toward lithium-ion. The case study will analyze moderate and high temperature installations.
Table 3: Generic System Specifications
|
Energy Delivered |
50 kWh |
|
Discharge rate |
5 hours |
|
Cycle frequency |
1 cycle/day |
|
Average ambient temperature |
Moderate: 25°C /77°F Hot: 33°C/92°F |
|
System life span |
1,900 cycles/5.2 years |
The site preparation, delivery, and installation costs will be calculated based on a 5.6:1 volume ratio of the lead acid system compared to the lithium-ion. This number is based on the fact that lithium-ion has 3.5 times the energy density of VRLA and uses an 80% DoD range compared to 50% for VRLA. VRLA is used in this analysis because it is a popular battery for grid storage and off-grid energy storage applications.
Based on the system life goal and rated cycle life of VRLA compared to lithium-ion, the VRLA system will have to be replaced once during the lifespan of the project for the moderate climate and three times for the hot climate. In both climates, the lithium-ion will not have to be replaced.
The lifetime cost, measured in $/kWh, is the critical number to understand the system economics. To calculate this, the sum of the battery, installation and transportation costs is multiplied by the number of times that a new system is required over the project period including the original install. The product of this multiplication is divided by the net energy throughput of the battery (50 kWh/cycle, 365 cycles/year, 5.2 years). Results are summarized in Table 4.
Table 4: Lifetime cost comparison of VRLA to Li-ion
|
VRLA (moderate climate: 25°C) |
VRLA (hot climate: 33°C) |
Lithium-ion |
|
|
System size |
100 kWh |
100 kWh |
62.5 kWh |
|
Battery Cost |
$12,000 ($120/kWh) |
$12,000 ($120/kWh) |
$37,500 ($600/kWh) |
|
Cycle Life |
1,000 @ 50% DoD |
500 @ 50% DoD |
1,900 @ 80% DoD |
|
Installation |
$20/kWh |
$20/kWh |
$3.6/kWh |
|
Transportation |
$28/kWh |
$28/kWh |
$5/kWh |
|
Lifetime cost |
$0.34/kWh throughput |
$0.67/kWh throughput |
$0.40/kWh throughput |
The analysis indicates that lithium-ion has an 18% higher lifetime cost when compared to VRLA in moderate climates, but is much more cost effective in hot climates. Based on Figure 11, there is a significant area of the world that sees average temperatures high enough to decrease the life of lead acid batteries. A factor not represented in the figure is that the battery systems are often housed in enclosures that see internal temperatures 10°C higher than the air temperature due to solar insolation, which would further decrease the performance of lead acid. The average temperature is also not completely representative of how much time is spent at extreme temperatures where the degradation accelerates in lead acid systems (e.g. one hour spent at 40°C and one hour spent at 20°C has a worse impact on the battery compared to two hours spend at 30°C).

Figure 11: Annual Global Temperature[iv]
-
Conclusions
Lead acid and lithium-ion offer pros and cons for the stationary energy storage industry. When calculating the balance of the pros and cons for a specific application, many factors must be considered. The importance of initial cost, lifetime, weight, volume, temperature sensitivity, maintenance access and access to product all play a role in battery selection.
There is currently no single solution that is better than another for all situations, but lithium-ion systems offer better cost of ownership in a number of situations, specifically hot climates, despite the higher initial cost.
[1] There is a wide price range for lithium-ion. $600/kWh represents estimated price at moderate production volumes. A similar concept applies to lead acid. There are various quality levels of lead acid, so some lead acid may have better cycle life, but will have a higher cost.
[i] Al-Hallaj S, Selman J. Thermal modeling of secondary lithium batteries of electric vehicle/hybrid electric vehicle applications. J Power Sources. 2002: 110; 341-348.
[iii] Environ. Sci. Technol. 2010, 44, 6550–655
[iv] Robert A. Rohde for Global Warming Art.
The content & opinions in this article are the author’s and do not necessarily represent the views of AltEnergyMag
Comments (2)
Featured Product


