The Future of Bio-Inspired Batteries
By Len Calderone for, Altenergymag
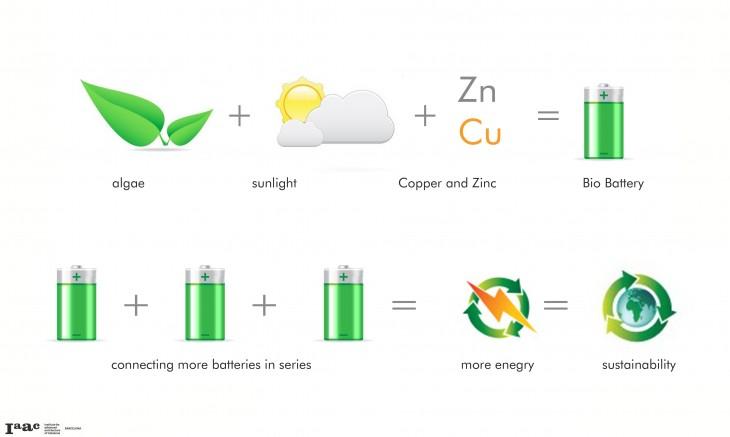
Scientists have investigated how basic polymer and protein building blocks that exist in nature can be used as guides to create hybrid materials. This is a new field in bioelectronics, where self-assembling proteins can be used as the basis for creating three-dimensional organic structures that can be used for electronic components. These materials can lead to new designs in bio-inorganic devices for energy storage, catalysis, solar cells and fuel cells.
Using nature as a guide, new materials can be created, or the natural material itself can be utilized. By learning how the molecular components of living objects link, we can duplicate the same structures to build materials of our own.
Organic materials, such as plastics, diamonds and biological proteins, can be combined with inorganic materials, which are used in the electronics industry to help solve our need for more energy efficiency. The combination of organic and inorganic materials can be used to improve battery storage capacity and energy collecting.
Let's look at how a battery is comprised. There are three main parts—two electrodes with opposite charges and an electrolyte that is located between the two. One of the electrodes is called the cathode. The cathode is made of two types of material that need to be in close proximity to one another so that the electrons can effectively flow between them.
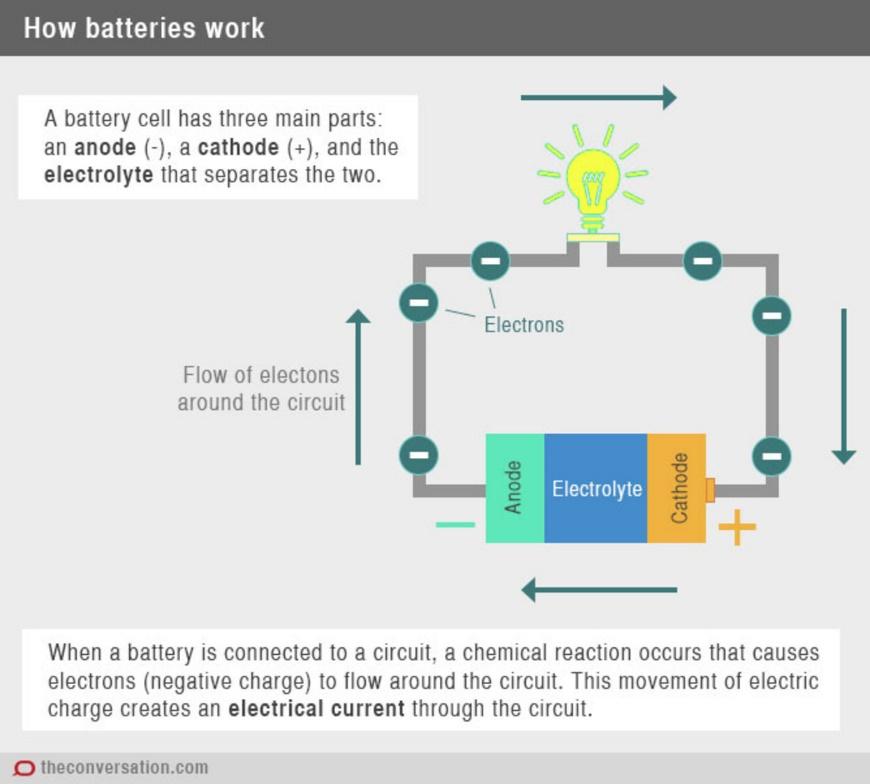
One of the materials must have high electro-activity, allowing the lithium ions in the electrolyte to quickly insert themselves into and remove themselves from its chemical structure. Lithium ions move from the negative electrode to the positive electrode during discharge and back when charging. Combining electroactive and conductive materials improves the performance of the battery. The electrolyte, which allows for ionic movement, and the two electrodes are the constituent components of a lithium-ion battery cell.
Better batteries with less expensive materials could result in more electricity from renewable energy sources by storing large amounts of energy until needed. Scientists think they may have the solution, with the help of a little organic chemistry. They turned to quinones, a carbon-based class of chemicals, which move electrons in the chemical reactions we use to store energy in our bodies. Researchers suspected that quinones also could move electrons in flow batteries.
In the January 2014 journal, Nature, researchers described a prototype flow battery that uses AQDS for one electrode. They showed that not only does it hold more energy than a conventional flow battery, but the cost of the quinone battery is much lower than other flow batteries.
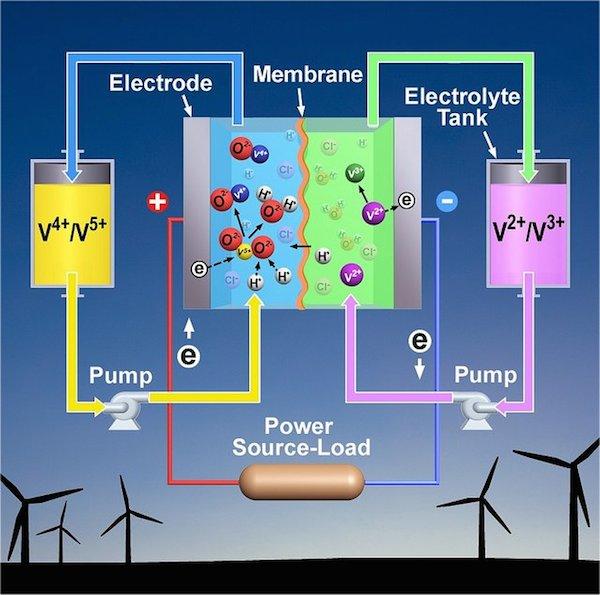
Like conventional batteries, flow batteries store chemical energy and have a positive and negative electrode. When the battery is discharged, electrons and ions move from one electrode to the other, generating a current. Conventional battery materials are solid, while a flow battery's chemicals are liquid.
Scientists are looking to nature for a breakthrough that will enhance the electrochemical performance of lithium-ion batteries. The researchers have developed tiered porous carbon spheres to be used as anodes.
In nature, a large number of microorganisms, like diatoms, can assemble bio-minerals into intricate tiered three-dimensional architectures with great structural control. These organisms contain organic macromolecules, which can be used as templates to induce and direct the precise precipitation of silica building blocks to form the complex structures to produce carbon materials for use as anodic components of batteries.
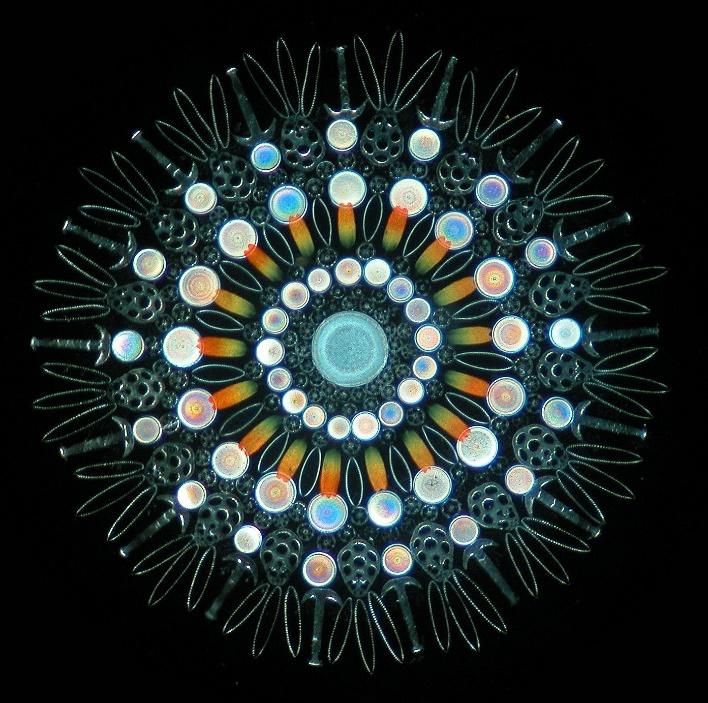
Diatom
These materials contain mesopores, which form an interconnected network of channels within the carbon spheres, and have a microporous surface. These three-dimensional features promote ion transport and high storage capacity within the carbon spheres.

Mesopores
So, what does all of this mean? Batteries contain heavy metals and other materials that are toxic to the environment. Bio-inspired battery technology, or bio-batteries, overcomes many of these problems. In the future, this technology may lead to biodegradable batteries that produces energy more efficiently than today's heavy duty lithium-ion batteries.
Because lithium technology is dangerous, the protective container takes up a lot of space. The protective cases are limiting battery space. As electronics continue to improve, we will need higher battery capacities to power them. Therefore, we will be designing batteries so that they can be used for the applications that we require.
We are seeing batteries being developed that are for all intents and purpose edible—not that we eat them. These batteries are inspired by biology since every day we consume food, and the cells in our bodies convert that food into energy.

The energy conversion component of a living cell happens in the mitochondria. Therefore, the mitochondria are removed intact from yeast, spinach, or potatoes and put on an electrode surface; or the part of the actual enzymes or catalysts in the mitochondria that do energy conversion from the cell are removed and put on the electrode surface.
The fuel for a bio-inspired battery is not a dangerous chemical, but are the same kinds of things that we ingest, like sugar or alcohol. The mitochondria or enzymes convert the fuel into electrical energy. This technology is extremely efficient in the living cell, and if we can get that kind of efficiency we would have energy densities that are roughly twenty times as energy dense as lithium-ion batteries.
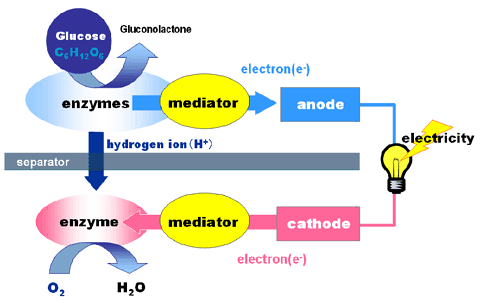
The disposal of bio-inspired batteries has apparent advantages, as Americans throw away 358 million pounds of batteries ever year, dispersing toxic materials into our air, water, and soil. There could be other benefits as well, such as a pacemaker that runs on a person's blood sugar; or the batteries could be make flexible for use as wearable electronics.

A major advantage that bio-inspired batteries have as compared to other batteries is their ability to allow an instant recharge. By utilizing a constant supply of sugar, or glucose, bio-inspired batteries are able to continuously keep themselves charged without an external power supply. Bio-inspired batteries are also a source of non-flammable, and non-toxic fuel. This provides a clean alternative renewable power source.
Bio-inspired batteries have a promising future ahead of them as tests and research have increased of late. Bio-batteries indicate to be a new form of energy that is proving to be environmentally friendly. More tests will need to be run before they are commercially sold.
For more information:
-
http://faculty.chemistry.harvard.edu/files/gordon/files/quinone_electrochem_acs_2015.pdf
-
http://santafe.edu/media/cms_page_media/500/BioEnergyAndreaKristenRevised_10-06-2013.pdf
Len Calderone - Contributing EditorLen contributes to this publication on a regular basis. Past articles can be found in the Article Library and his profile on our Associates Page He also writes short stores that always have a surprise ending. These can be found at http://www.smashwords.com/profile/view/Megalen. |
 |